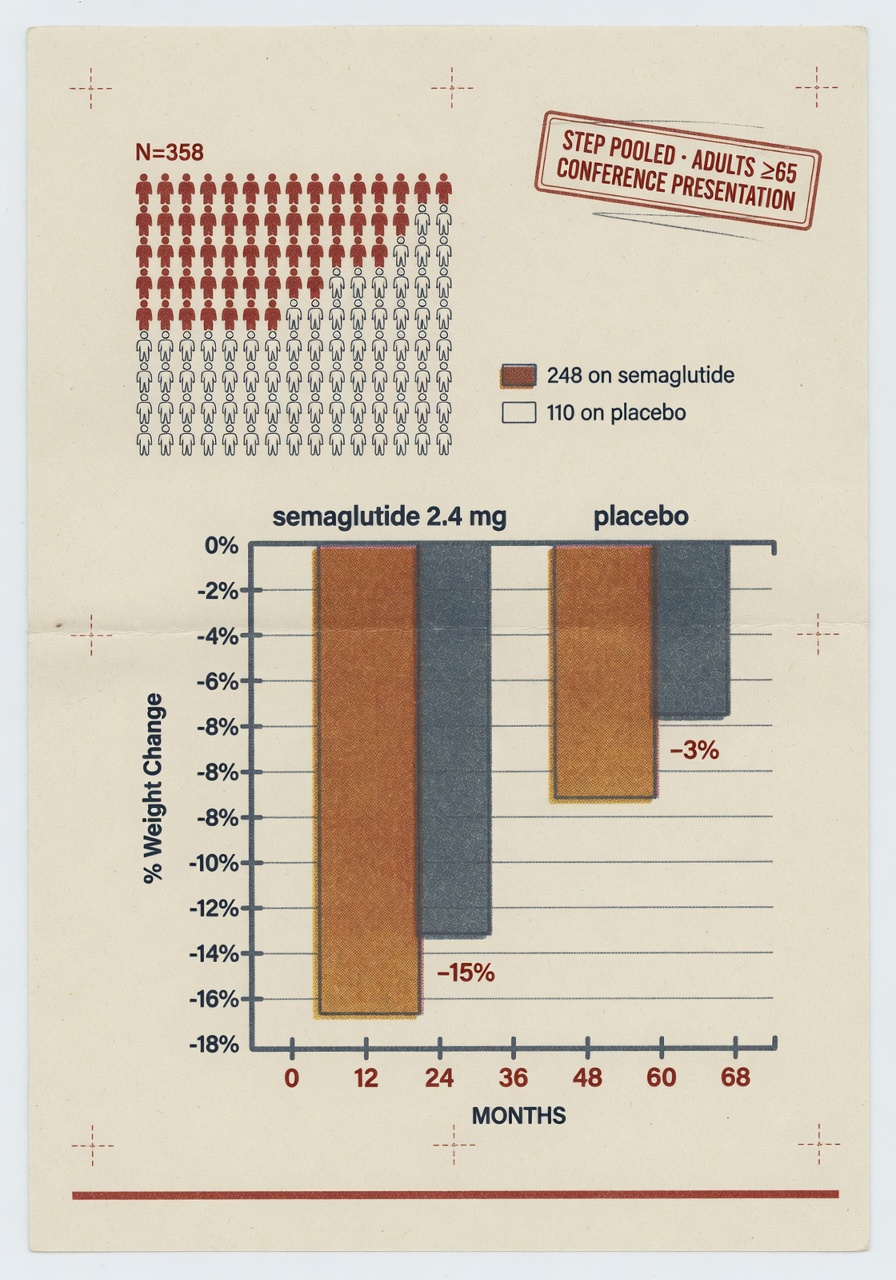
A pooled analysis of six STEP trials, presented on Wednesday at the European Congress on Obesity in Istanbul, reports that adults aged 65 and over with obesity but without diabetes lost a mean 15.4 percent of body weight on once-weekly subcutaneous semaglutide 2.4 mg over 68 weeks, against 5.1 percent on placebo. The subgroup is 358 participants drawn from the STEP 1, 3, 4, 5, 8, and 9 trial populations (semaglutide arm, 248; placebo, 110), about 8 percent of the 4,523-person pool. The work was led by Luca Busetto of the University of Padova, funded by Novo Nordisk, and assigned conference abstract 1681. It is a conference presentation, not a peer-reviewed paper, and the numbers should be read with that label attached.
The figure that drove the wires this week, mean weight loss of 15.4 percent versus 5.1 percent at 68 weeks, is consistent with what STEP 1 reported in 2021 for the full trial population (14.9 percent versus 2.4 percent at 68 weeks, New England Journal of Medicine, March 2021). Within the over-65 subgroup, 66.5 percent of semaglutide participants achieved a weight reduction of at least 10 percent, 46.8 percent at least 15 percent, and 28.6 percent at least 20 percent; the placebo arm reached the same thresholds at 15.5 percent, 6.4 percent, and 2.7 percent respectively. Mean waist-circumference reduction was 14.3 cm against 6.0 cm. These are the numbers Novo Nordisk released; the conference abstract has been distributed but the full data tables remain congress-restricted until publication.
What the subgroup represents, and what it does not
The STEP trials enrolled adults with a body-mass index of 30 or higher, or 27 or higher with at least one weight-related comorbidity, and excluded participants with type 2 diabetes; the pooled analysis carries those entry criteria forward. Participants in the subgroup had a mean age of 69 years and were 72.3 percent women. STEP trial protocols also exclude participants with prior bariatric surgery, recent significant weight loss, and several active medical conditions; this is the standard envelope for a phase-three obesity-drug trial. The clinical population over 65 with obesity in the United States and Europe is broader than that envelope. The Centers for Disease Control and Prevention reports obesity prevalence of 38.4 percent among U.S. adults aged 60 and over in the 2017–March 2020 National Health and Nutrition Examination Survey cycle, and the largest single contributor to that figure is people with concurrent type 2 diabetes — the population the STEP trials explicitly screened out.
A 358-participant subgroup is small for inference about an age band. The pooled analysis collapses six trial populations to gain statistical power on age but does so at the cost of heterogeneous comparator regimens, lifestyle-intervention intensities, and follow-up windows across the constituent trials. STEP 1 ran 68 weeks against placebo and lifestyle counselling; STEP 4 examined withdrawal from semaglutide after a 20-week run-in; STEP 5 ran for two years; STEP 8 compared semaglutide with liraglutide. Pooling these populations is methodologically defensible for the precise endpoint reported (mean weight change at the 68-week mark), but the resulting subgroup is not a clean cohort, and the conference abstract will need full peer review before the underlying analytic choices are visible to outside readers.
Safety: similar profile, comparable serious-event rate
The safety figures reported with the analysis are the second piece of news, and they carry their own caveats. Gastrointestinal adverse events were the dominant tolerability signal in the over-65 arm, as they are in the broader STEP populations; rates of constipation and dizziness were higher than in the general trial population, the abstract reports. Serious adverse events were recorded in 19.0 percent of the semaglutide arm against 12.7 percent of the placebo arm, a 6.3-percentage-point gap that the authors describe as comparable to the general trial population. Whether that gap reflects drug-attributable events or the higher background event rate expected in any older cohort is a question the abstract does not resolve, and the answer requires the full event-level data.
Two safety questions that matter for older adults are not addressed in this subgroup analysis and have not been addressed in the broader STEP trial program either. The first is sarcopenia. Substantial weight loss in older adults is accompanied by loss of lean mass and bone mineral density; the STEP body-composition substudy (STEP 1 DXA arm, 140 participants, mean age 47 years) found about 40 percent of weight lost came from lean mass, but the sub-cohort was too young and too small to characterise the effect in the 65-and-over band. The second is muscle-function and fall-risk endpoints. Neither the STEP trials as a class nor this pooled analysis carries pre-specified muscle-strength, gait-speed, or fall-rate outcomes. The Veterans Affairs SEMA-RX-OA randomised trial currently enrolling at five U.S. sites is designed to address those gaps and is not expected to read out until 2028.
When you treat people aged 65 years and older, you can expect similar results to younger people in terms of weight loss, metabolic improvement, and side effects. — Luca Busetto, professor of nutrition and dietetic sciences, University of Padova
What the result changes for clinical practice, and what it does not
The Medicare prescription-drug benefit has covered semaglutide for obesity in beneficiaries with established cardiovascular disease since March 2024, when the Centers for Medicare and Medicaid Services issued a memorandum interpreting the SELECT trial result (NEJM, November 2023) as bringing semaglutide within the Part D definition of a covered drug when prescribed for a medically accepted indication beyond weight loss alone. The STEP pooled analysis does not change that coverage posture; it does not establish a new indication, and the over-65 subgroup includes participants without cardiovascular disease, who would not currently qualify for Part D coverage on the cardiovascular pathway. The American College of Cardiology and American Diabetes Association have not yet revised their position statements on GLP-1 receptor agonist use in older adults to reflect this analysis, and would not be expected to before peer-reviewed publication.
What the analysis does is narrow the inferential gap. Before this week, the clinical case for prescribing semaglutide to a 70-year-old patient with obesity rested on extrapolation from trials whose mean age sat in the high forties. The pooled subgroup now offers a within-trial efficacy estimate for the over-65 band that lines up with the broader STEP results. That is useful. It is not the same as a dedicated trial powered on geriatric endpoints, and it does not speak to the populations the STEP entry criteria excluded — adults with diabetes, with prior bariatric surgery, with recent significant weight loss, or with the comorbid burdens that bring most patients in this age range into the obesity-medicine office in the first place.
What to watch
Three follow-on items are worth tracking by name. First, the peer-reviewed manuscript: Busetto and colleagues have indicated a submission is in preparation but have not named a target journal; until it appears and the analytic choices are visible, the 15.4 percent figure is congress-data, not literature-data. Second, the Novo Nordisk STEP HFpEF follow-on (NCT05371496) and the SELECT prespecified older-adult analysis, both of which will speak to the cardiovascular-event question in over-65 patients with established disease. Third, the VA SEMA-RX-OA trial, which is the first randomised study in this drug class to carry pre-specified sarcopenia, gait-speed, and fall outcomes. Until those read out, the prescribing decision in older adults turns on a 358-person subgroup whose strongest claim is consistency with the broader STEP record, not independence from it.